The #ImpatientRevolution eBook
This eBook is a guide for impatient patient organizations – those wanting to accelerate the development of new medicines for their disease by taking an active role. It explains why and how companies develop drugs for rare diseases, and how patient organizations can use this knowledge to analyze their disease field, to identify the main gaps, and to design strategies to get pharmaceutical companies to develop new treatments for their disease.
“#ImpatientRevolution is a free digital download eBook which is a must-read for charities about to embark on the journey towards drug development and clinical trials. Packed full of practical advice and tools, it’s a useful reference to guide you through the process.”
Summaries from conferences
The American Epilepsy Society (AES) meeting is the largest epilepsy meeting of the year, and because it takes place every month of December it also serves as an annual review on the understanding and treatment of epilepsies. These are my top 5 insights from the American Epilepsy Society 2023 meeting.
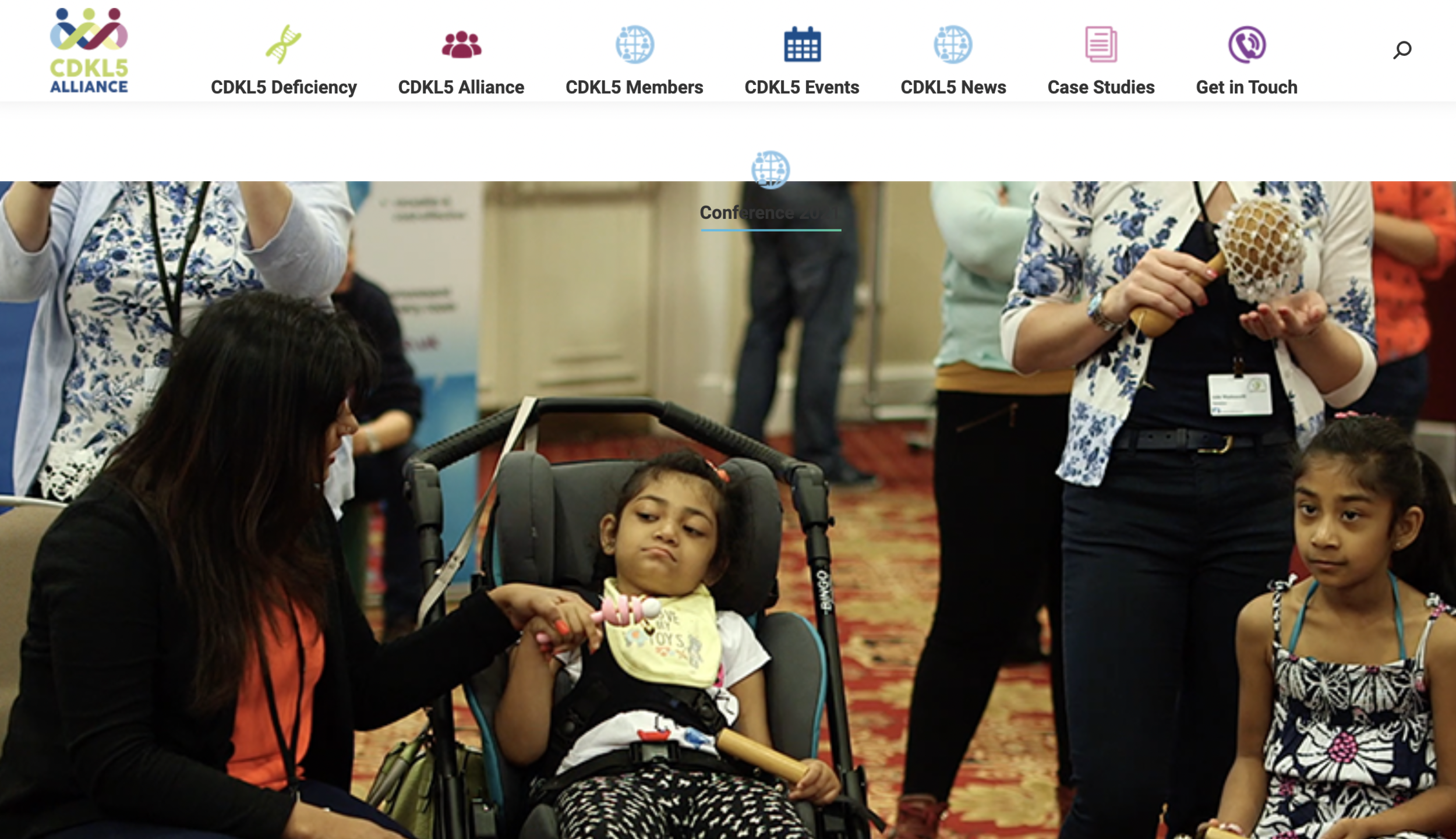
La novena edición del Foro CDKL5 tuvo lugar en Boston, los días 6 y 7 de noviembre. El Foro es una reunión anual que organiza la Fundación Loulou y en la que científicos y miembros de la industria farmacéutica se reúnen con representantes de la comunidad de pacientes para repasar los últimos avances en el campo.
Este es un repaso para los grupos de pacientes de las principales novedades del Foro CDKL5 2023.
For the past nine years the Loulou Foundation hosts an annual meeting where scientists and drug developers working on CDKL5 deficiency, together with representatives from patient organizations, meet to discuss the latest advances.
Here are the main news and take-home messages from the 2023 CDKL5 Forum that took place in November 6-7 2023
Una conversación que suele salir mucho en reuniones de familias CDKL5 es la posición de la mutación de sus hijos en el gen, por si eso nos ayuda a saber la severidad de la enfermedad cuando crezcan. Os resumo abajo un listado de publicaciones y sus conclusiones.
The International Epilepsy Congress (IEC) is one of the largest epilepsy meetings attracting clinicians, researchers and the pharmaceutical industry. In 2023 it took place in Dublin, in early September. I review the progresses towards treating the cause of the rare epilepsies / DEEs
The American Epilepsy Society (AES) meeting is the largest epilepsy meeting of the year, and because it takes place every month of December it also serves as an annual review on the understanding and treatment of epilepsies. I review the progresses towards treating the cause of the rare epilepsies / DEEs.
For the past eight years the Loulou Foundation hosts an annual meeting where scientists and drug developers working on CDKL5 deficiency, together with representatives from patient organizations, meet to discuss the latest advances.
Here are the main news and take-home messages from the 2022 CDKL5 Forum that took place in November 7-8 2022.
La octava edición del Foro CDKL5 tuvo lugar en Boston, los días 7 y 8 de noviembre. El Foro es una reunión anual que organiza la Fundación Loulou y en la que científicos y miembros de la industria farmacéutica se reúnen con representantes de la comunidad de pacientes para repasar los últimos avances en el campo.
Este es un repaso para los grupos de pacientes de las principales novedades del Foro CDKL5 2022.
The American Epilepsy Society (AES) meeting is the largest epilepsy meeting of the year, and because it takes place every month of December it also serves as an annual review on the understanding and treatment of epilepsies. These are my top 5 insights from the American Epilepsy Society 2021 meeting.
For the past seven years the Loulou Foundation hosts an annual meeting where scientists and drug developers working on CDKL5 deficiency, together with representatives from patient organizations, meet to discuss the latest advances.
Here are the main news and take-home messages from the 2021 CDKL5 Forum that took place in November 1-2 2021.
CDKL5 deficiency is biologically reversible, bringing much hope and creating a sense of urgency for the development of restorative treatments for CDKL5 deficiency disorder (CDD). These are the conclusions of a publication from Zhaoland (Joe) Zhou’s lab from the University of Pennsylvania. The study was just published in the Journal of Clinical Investigation, and it is poised to become a landmark study in the field.
Last weekend, the CDKL5 deficiency disorder (CDD) community gathered in front of their computers for a virtual annual meeting organized by the International CDKL5 Alliance. For the 2021 Alliance meeting, the organizing team commission a series of pre-recorded videos to a large range of speakers and published the videos with subtitles so that all CDD families from all around the world could watch them at the same time. You can also access the videos at the CDKL5 Alliance website. Here is a personal summary of the 2021 CDKL5 Alliance meeting.
As of end 2020, the Dravet syndrome pipeline comprises 3 approved drugs, 11 drug candidates, and 12 different products have received orphan drug designations. Since the last report the pipeline has changed by, among others, the approval by FDA and EMA of Epidyolex (cannabidiol) and Fintepla (fenfluramine), the initiation of the first clinical trial with an antisense therapy, and progresses in different disease-targeting modalities including gene therapy approaches.
The American Epilepsy Society (AES) meeting is the largest epilepsy meeting of the year, and because it takes place every month of December it also serves as an annual review on the understanding and treatment of epilepsies. This year the meeting was virtual, which made the exhibit hall and the poster sessions less exciting but also made the presentations more accessible, including a 90-day on-demand period after the meeting to catch up with all of the parallel tracks. These are my own 5 insights from the American Epilepsy Society 2020 meeting.
For the past six years the Loulou Foundation hosts an annual meeting where scientists and drug developers working on CDKL5 deficiency, together with representatives from patient organizations, meet to discuss the latest advances. This was the fourth Forum I attended, and my third since joining the Loulou Foundation.
Here are the main news and take-home messages from the 2020 CDKL5 Forum that took place in October 12-14 2020.
La sexta edición del Foro CDKL5 tuvo lugar online, los días 12-14 de octubre. El Foro es una reunión anual exclusivamente por invitación que organiza la Fundación Loulou y en la que científicos y miembros de la industria farmacéutica se reúnen con representantes de la comunidad de pacientes para repasar los últimos avances en el campo. Este ha sido mi cuarto Foro CDKL5, y el tercero desde que me uní a la Fundación Loulou.
Este es un repaso para los grupos de pacientes de las principales novedades del Foro CDKL5 2020. [SPANISH VERSION - ALSO AVAILABLE IN ENGLISH]
There are multiple gene therapy and oligonucleotide programs in development for Dravet syndrome including those that supply and extra copy of the SCN1A gene and those that boost expression from the healthy SCN1A gene copy. Clinical trials have already started, with Stoke Therapeutics initiating the first clinical trial with a disease-targeting therapy in Dravet syndrome in summer 2020. Behind Stoke, gene therapies are approaching the clinic with Encoded Therapeutics having the most advanced clinical candidate and preparing for trials in 2021.
The rare genetic disease CDKL5 Deficiency Disorder (CDD) has been designated with a new disease code in the International Classification of Diseases (ICD), the medical classification list from the World Health Organization (WHO). The CDKL5 Deficiency Disorder diagnostic code will be incorporated in the October 1, 2020 classification revision.
This article covers what is an ICD-10, why it is important, and how patient communities can apply to have a code created for their disease.
Cada vez que se anuncia un nuevo ensayo clínico los pacientes y sus familias experimentan una mezcla de emociones. De un lado la alegría y esperanza de saber que vienen nuevos fármacos para su enfermedad. Del otro lado, muchas veces, desilusión y sensación de injusticia al ver que muy posiblemente no tengan la oportunidad de participar en el ensayo. Como científico involucrado en el diseño y ejecución de ensayos clínicos quiero compartir con los pacientes y sus familias algunas notas sobre por qué no todos los pacientes pueden entrar en un ensayo clínico, y si esto es “justo” o “injusto”.
El congreso de la Sociedad Americana de Epilepsia (AES por sus siglas en ingés) es la mayor cita en epilepsia del año, y como tiene lugar cada mes de Diciembre sirve también para hacer un repaso de como ha avanzado el campo a lo largo del ultimo año. AES 2019 fue el año de las terapias genéticas para las encefalopatías epilépticas y de desarrollo. Este artículo es el resumen de lo que me pareció mas interesante del congreso AES 2019
The American Epilepsy Society (AES) meeting is the largest epilepsy meeting of the year, and because it takes place every month of December it also serves as an annual review on the understanding and treatment of epilepsies. AES 2019 was the year of genetic therapies for the developmental and epileptic encephalopathies. This article highlights what I found the most interesting at the AES 2019 meeting.
La quinta edición del Foro CDKL5 tuvo lugar en Boston, los días 4 y 5 de noviembre. El Foro es una reunión anual exclusivamente por invitación que organiza la Fundación Loulou y en la que científicos y miembros de la industria farmacéutica se reúnen con representantes de la comunidad de pacientes para repasar los últimos avances en el campo. Este ha sido mi tercer Foro CDKL5, y el segundo desde que me uní a la Fundación Loulou.
Este es un repaso para los grupos de pacientes de las principales novedades del Foro CDKL5 2019. [SPANISH VERSION - ALSO AVAILABLE IN ENGLISH]
For the past five years the Loulou Foundation hosts an annual meeting where scientists and drug developers working on CDKL5 deficiency, together with representatives from patient organizations, meet to discuss the latest advances. This was the third Forum I attended, and my second since joining the Loulou Foundation.
Here are the main news and take-home messages from the 2019 CDKL5 Forum that took place in Boston in November 4 and 5.
The 2019 Dravet Syndrome Pipeline and Opportunities Review provides a review and analysis of 12 drug candidates in development for the treatment of Dravet syndrome, including 11 products that have received orphan drug designations. The Report includes the most recent updates on programs from GW Pharmaceuticals (Epidiolex / Epidyolex), Zogenix (Fintepla, ZX008), Biocodex (stiripentol) , Ovid Therapeutics (Soticlestat, OV935, TAK-935), Takeda Pharmaceutical, Supernus Pharmaceuticals (SPN-817, Huperzine), Xeris Pharmaceutical (diazepam), Epygenix Therapeutics (EPK-100, -200 and -300), NeuroCycle Therapeutics (NCT10015), PTC Therapeutics (ataluren), Stoke Therapeutics (STK-001), Encoded Therapeutics and OPKO Health (OPK88001, CUR-1915).
I used to say that at the patient communities “we set the agenda”. It turns out we didn’t, we were borrowing the agenda from scientific meetings. The 2019 CDKL5 Alliance International Research and Family Conference redefined what a patient-centered conference truly is. In this article I summarise the elements that make a meeting truly patient-centered.
There are currently 4 clinical trials ongoing or about to start in CDKL5 Deficiency Disorder: ataluren, ganaxolone, TAK-935 and fenfluramine. This article is a summary of where we are with clinical trials for CDKL5 Deficiency Disorder for families and other interested readers including what we know about these four drugs, their efficacy, at which level of clinical development they are at, and where can you learn more about these trials.
Many orphan drugs are advanced therapies. Pricing and access are major issues. Epilepsy is catching up with gene therapy. We shouldn’t call them rare diseases, but frequently misdiagnosed diseases. Either we wait 2,000 years for treatments or we start thinking “many diseases at a time”, and online patient communities are now part of the drug development process. That’s the short summary of the main lessons I took home from attending the World Orphan Drug Congress at the National Harbor April 10-12. The WODC one of the largest meetings dedicated to the development of new medicines for rare diseases and takes place once in the US and once in Europe every year. In a bit more detail, here is the expanded list of what I would like to share with you from the conference.
There are multiple gene therapy programs in development for Dravet syndrome including those that supply and extra copy of the SCN1A gene and those that boost expression from the healthy SCN1A gene copy. Clinical trials are around the corner, with Stoke Therapeutics expecting to initiate clinical trials in 2020. Just Stoke is not enough. New corporate players, and ideally some precompetitive collaboration around the common challenges of validating clinical outcome measures and biomarkers, are needed to maximize the success of gene therapies for Dravet syndrome.
2019 will be the year when we might have the European launch of Epidiolex, the US approval and launch of Fintepla, an ongoing clinical trial with TAK-935, hopefully some news about the ability of Translarna to improve Dravet syndrome by rescuing some of the nonsense mutations, and a year to prepare for the clinical trials that starting in 2020 will dominate the field: gene therapy approaches for Dravet syndrome that will treat more than just seizures. This entry reviews when we expect the main news about the Dravet syndrome pipeline during 2019.






























These are my main learnings from the Syngap Conference 2023 celebrated in Orlando the day before the American Epilepsy Society meeting.